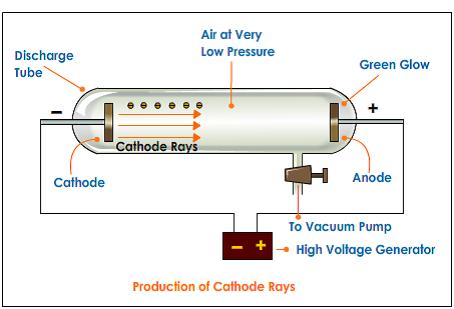 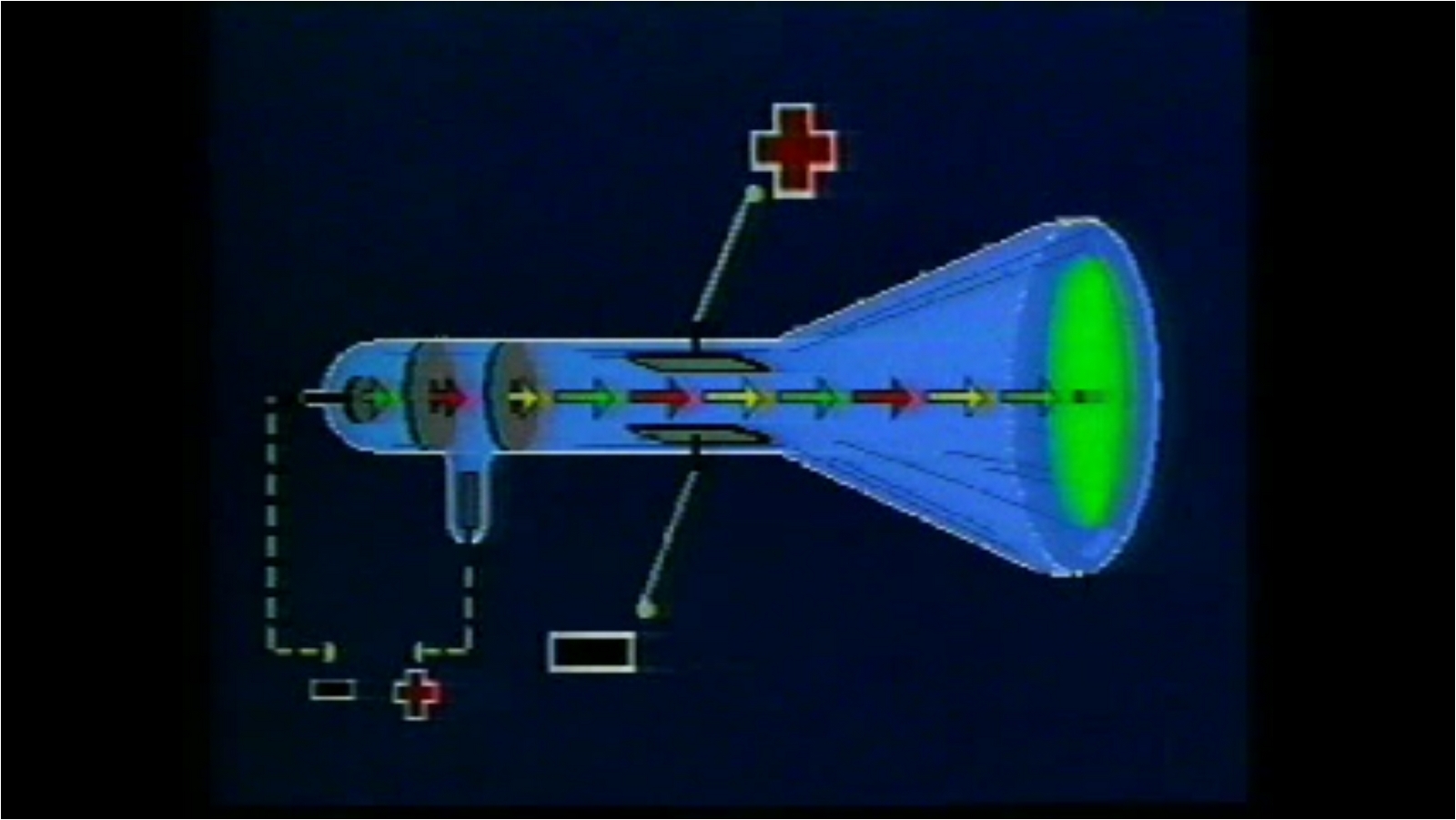
To release electrons into the tube, they first must be detached from the atoms of the cathode. The Maltese cross has no external electrical connection.Ĭathode rays are so named because they are emitted by the negative electrode, or cathode, in a vacuum tube. Cathode-ray tubes (CRTs) use a focused beam of electrons deflected by electric or magnetic fields to render an image on a screen.Ī diagram showing a Crookes tube connected to a high voltage supply. 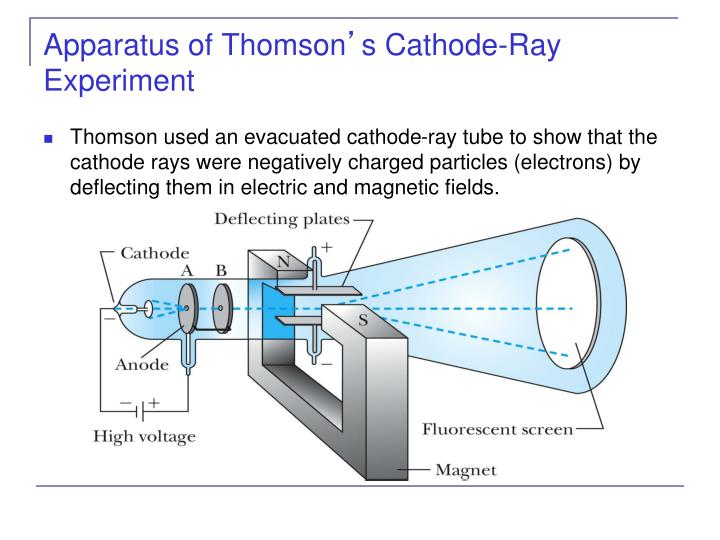
Thomson showed that cathode rays were composed of a previously unknown negatively charged particle, which was later named the electron. They were first observed in 1859 by German physicist Julius Plücker and Johann Wilhelm Hittorf, and were named in 1876 by Eugen Goldstein Kathodenstrahlen, or cathode rays. If an evacuated glass tube is equipped with two electrodes and a voltage is applied, glass behind the positive electrode is observed to glow, due to electrons emitted from the cathode (the electrode connected to the negative terminal of the voltage supply). Cathode rays are normally invisible in this demonstration Teltron tube enough gas has been left in the tube that the gas atoms luminesce when struck by the fast-moving electrons.Ĭathode rays or electron beam ( e-beam) are streams of electrons observed in discharge tubes. A beam of cathode rays in a vacuum tube bent into a circle by a magnetic field generated by a Helmholtz coil.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
